BeNa Culture Collection
-
Login |
-
EngLish

 info@bncc.com
info@bncc.com
BeNa Culture Collection

 info@bncc.com
info@bncc.com


| Culture medium | 293T/17 Cell Special Medium: 88% DMEM-H + 10% FBS + 1% Glutamine + 1% NEAA -BNCC |
| Subculture procedure | Recovery steps: ① The frozen vial is taken out of liquid nitrogen or -80 ℃ refrigerator and put into PE gloves, quickly submersed into a 37 ℃ water bath pan, shaken The frozen vial to accelerate dissolution, and it is advisable to dissolve all within 1min; (2) Add the dissolved cell solution into a centrifuge tube containing 9ml of complete culture medium in an ultra-clean table, centrifuge at 1000-1200rpm for 5min, discard the supernatant, resuspend the cells with 1-2ml of complete media. (3) The cell suspension was added to T25 flask containing 5-6mL complete medium and cultured in an incubator. cell subculture: ① remove the medium, rinse twice with PBS, and add 1-2mL pancreatin (0.25% Trypsin + 0.02% EDTA); (2) Observe the digestion situation under the microscope. When the cell edge shrinks and the adherent is loose (a pipette can be used to suck up some pancreatin and gently blow somewhere in the cell layer, and the cell layer can be seen to detach with naked eyes, I .e. digestion is completed, otherwise digestion is continued). directly suck out pancreatin, add 5-6mL of complete medium, gently blow the cell layer off. (3) dispense the cell suspension into a fresh T25 flask as a ratio of 1:2, add appropriate complete culture medium, mix the cell suspension evenly, and culture in an incubator.; ④ Pay attention to the change of pH of medium and cell density, renew the medium regularly (2-3 times a week), and repeat the subculture or cryopreservation when the cell density reaches 80%-90%. |
| Growth conditions | 37 ℃;5% CO2 + 95% air; |
| Growth characteristics | Adherent growth |
| Storage conditions | Liquid nitrogen |
| Safety level | 0 |
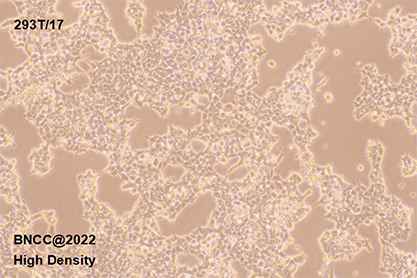
| morphology | Epithelial cell-like, spindle-shaped, irregular margin, monolayer adherent growth, clean background, no pigment production, no vacuoles |
| Sharing mode | Public welfare sharing |
1. Name:293T/17
2. No.:340228
3. Growth properties : ■ adherence to the wall □ suspension □ semi-suspension and semi-adherence
4. Growth conditions:
| culture medium | 90% & nbsp; High sugar DMEM |
| serum | 10% FBS (Diagnovum) |
| temperature | 37 ℃ |
| air condition | 5% CO2,95% AIR |
| growth algebra | P4-5 |
| frozen storage conditions | culture medium 50%, serum 40%, DMSO 10% |
5. Composition:
| composition | specifications |
| a bottle of cells | T25 |
| cell culture and operating instructions | 1 copy |
Receiving notice:
1 Upon arrival, it is suggested to sit the cells in a incubator for about 4 hours, and then renew the media for recovery or subculture according to the cell density.
2 If the adherent cells are received in the form of ( partial) suspension, please centrifuge the suspended cells in time, add 15% serum complete medium to a fresh culture dish / vial and continue to culture for 3 days; At the same time, the remaining adherent cells in the original culture flask were renewed with 15% serum complete medium and cultured for 2-3 days. If the cells do not proliferate after 3 days but continue to detach and die, please contact the technicians.
3 Slow growth of adherent cells: properly increase the serum concentration (no more than 20%) or transfer to a fresh culture flask for further culture after trypsin digestion according to the cell density.
4 Uneven growth: if adherent cells grow unevenly and is island-like, they can be digested, dispersed, and cultured with fresh medium.
Recovery and subculture procedure ( under strict aseptic conditions )
1 Remove the medium in the original culture flask, rinse twice with PBS, and add 2~3 ml of 0.25% EDTA for trypsin digestion (usually in 1~2min)
2 Observe the digestion under the microscope. When the cell edge shrinks and adherent is loose (but not floating), remove the trypsin, add 6~8ml complete medium, aspirate the cell layer off.
3 Transfer part of the cell suspension to a fresh culture vessel / flask, add appropriate complete medium, and culture it in the incubator.
4 Pay attention to the change of pH value of medium and cell density, renew the medium regularly, and repeat the subculture or cryopreservation when the cell density reaches 70%-80%.
Special attention: (if handle in public laboratory or first time to culture the cells, it is recommended to add penicillin streptomycin into the media)
1 Upon the receipt of the cells, renew the solution with fresh 10% FBS medium as soon as possible. It is not recommended to use the medium used for transportation in the original bottle.
2 Please take photos and contact the technicians in time if the culture flask leaks upon the receipt of cells.
3 Any compliant on the cells, please take photos and contctat our technicians.
 MT-4(MT-4)
BNCC338052
MT-4(MT-4)
BNCC338052
 OCI-LY3(OCI-LY3)
BNCC338435
OCI-LY3(OCI-LY3)
BNCC338435
 HuT78(HuT78)
BNCC359882
HuT78(HuT78)
BNCC359882
![SW948[SW-948]](https://beinabncc.oss-accelerate.aliyuncs.com/News/d3e12572b0921eb522c87709e0880df6.jpg) SW948[SW-948](SW948[SW-948])
BNCC295321
SW948[SW-948](SW948[SW-948])
BNCC295321
 SU. 86.86(SU. 86.86)
BNCC360007
SU. 86.86(SU. 86.86)
BNCC360007
 U373(U373)
BNCC338603
U373(U373)
BNCC338603