BeNa Culture Collection
-
Login |
-
EngLish

 info@bncc.com
info@bncc.com
BeNa Culture Collection

 info@bncc.com
info@bncc.com
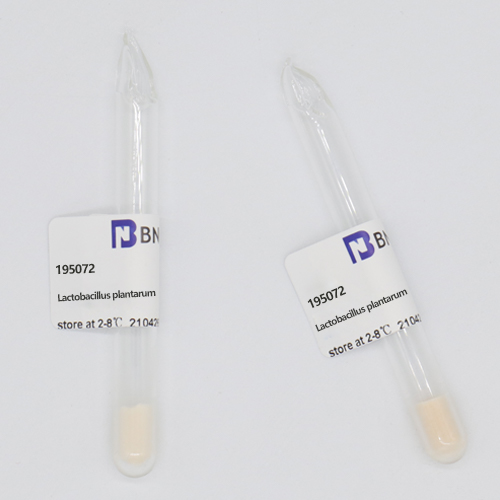


 Freeze-dried: lyophilized strain, freeze dried vial, used up at one time and not be retained
Freeze-dried: lyophilized strain, freeze dried vial, used up at one time and not be retained
| Culture medium | MRS medium (MRS): peptone 10.0g, beef paste 10.0g, yeast powder 4.0g, glucose 20.0g, magnesium sulfate 0.2g, sodium acetate 5.0g, triammonium citrate 2.0g, dipotassium hydrogen phosphate 2.0g, manganese sulfate 0.04g, Tween 80 1.0g, agar 20.0g (not included in liquid medium), distilled water 1.0L. pH 5.7±0.2. Sterilization at 121 ℃ for 15min. -BNCC |
| Subculture procedure | (1) Prepare a test tube containing 5~10mL of liquid medium and 2 plates; (2) Open it in the safety cabinet, heat the tip of ampoule in a flame, quickly drop sterile water to creak it, then break it with forceps; (3) draw 0.5mL of liquid culture medium into a freeze dried ampoule, fully rehydrate and transfer the solution to the liquid test tube, mix evenly; (4) inoculate a plate with 0.2mL of suspension liquid, repeat the step to obtain two plates; ⑤ Put all the liquid test tubes and plates under the above culture conditions for cultivation, and the strains can be used when they grow. |
| Growth conditions | 37 ℃;24-48h; Aerobic |
| Storage conditions | 2-8 ℃ |
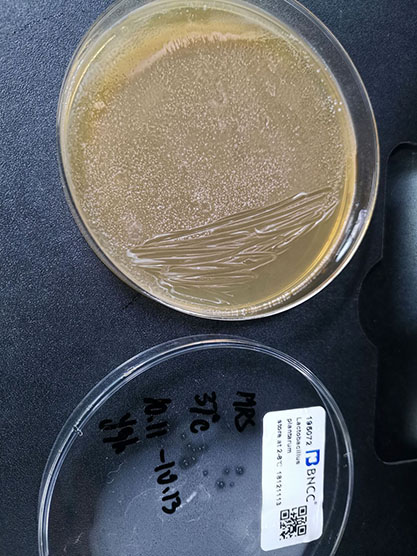
| morphology | Size: 1-2mm Shape: Round Edge: Neat Transparency: Opaque Color: milky white bulge: middle bulge surface: bright and smooth texture: moist and easy to stir up |
| Separation substrate | Yogurt |
| Sharing mode | Public welfare sharing |
1.Description
1.Name:Lactobacillus plantarum
2.BNCC No.:195072
3. Biosafety level:4
2.Storage conditions:
Storage of freezed dried ampoule and agar slant at 2°C to 8°C
3.Growth Conditions
1. MRS medium: peptone 10.0g, beef paste 10.0g, yeast powder 4.0g, glucose 20.0g, magnesium sulfate 0.2g, sodium acetate 5.0g, triammonium citrate 2.0g, dipotassium hydrogen phosphate 2.0g, manganese sulfate 0.05g, Tween 80 1.0g, agar 20.0g (not included in liquid medium), distilled water 1.0L. pH 6.2±0.2.
2. Atmosphere:micro aerobic
3. Temperature:37 ℃,2d
4.Notes:
1.Normal culturing time, 24-48hours for bacterial, 72 hours for yeast, 5-7days for mould, 7-10days for fungal.
2.Agar slant shall be inoculated asap, and do not keep the storage for more than 3 months.
3.Please recover the strains in strict accordance with this instruction, otherwise the replacement of the strain are not be available in case of viability loss caused by different media or growth conditions.
4.Waste generated from the handling process should be discarded after high-pressure sterilization
 Lactobacillus plantarum(Lactobacillus plantarum)
BNCC353606
Lactobacillus plantarum(Lactobacillus plantarum)
BNCC353606
 Lactobacillus plantarum(Lactobacillus plantarum)
BNCC194165
Lactobacillus plantarum(Lactobacillus plantarum)
BNCC194165
 Lactobacillus plantarum(Lactobacillus plantarum)
BNCC185343
Lactobacillus plantarum(Lactobacillus plantarum)
BNCC185343
 Lactobacillus plantarum(Lactobacillus plantarum)
BNCC191046
Lactobacillus plantarum(Lactobacillus plantarum)
BNCC191046
 Lactobacillus plantarum(Lactobacillus plantarum)
BNCC189746
Lactobacillus plantarum(Lactobacillus plantarum)
BNCC189746
 Lactobacillus plantarum(Lactobacillus plantarum)
BNCC187897
Lactobacillus plantarum(Lactobacillus plantarum)
BNCC187897