BeNa Culture Collection
-
Login |
-
EngLish

 info@bncc.com
info@bncc.com
BeNa Culture Collection

 info@bncc.com
info@bncc.com



| Subculture procedure | Resuscitation steps: ① Remove the frozen vial from liquid nitrogen or -80 ℃ refrigerator and place it in PE gloves. Quickly immerse it in a 37 ℃ water bath, shake the vial to accelerate dissolution, and dissolve it completely within 1 minute; ② Add the dissolved cell solution into a centrifuge tube containing 9mL of complete medium on a ultra-clean bench, centrifuge at 1000-1200rpm for 5 minutes, discard the supernatant, and resuspend the cells in 1-2mL of complete medium. ③ Add the cell suspension into a T25 flask containing 5-6mL of complete medium and place the flask in an incubator. Cell passage: ① Remove the medium, rinse twice with PBS , and add 1-2mL pancreatin (0.25% Trypsin + 0.02% EDTA); ② Observe the digestion situation under the microscope. When the cell edge shrinks and the adherent is loose (a pasteur pipette can be used to suck up some pancreatin and gently blow somewhere in the cell layer, and the cell layer can be seen to detach with naked eyes, I .e. digestion is completed, otherwise digestion is continued), directly suck out pancreatin, add 5-6mL of complete medium, gently blow the cell layer off. ③ Divide the cell suspension into a fresh T25 flask at a ratio of 1:2, add appropriate complete culture medium, mix the cell suspension evenly, and culture it in the incubator.; ④ Pay attention to changes in the pH value of the culture medium and cell density, change the medium regularly (2-3 times a week), and repeat the passage operation or cryopreservation when the cell density reaches 80% -90%. |
| Growth conditions | 37°C; 5% CO₂ + 95% air |
| Safety level | 0 |
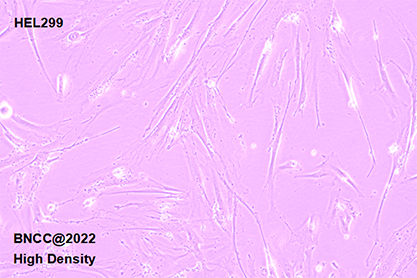
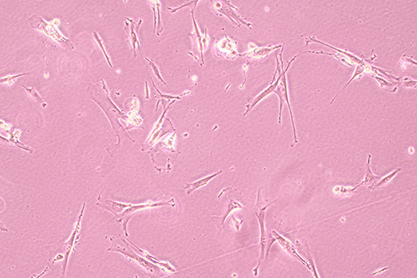
| morphology | Fibroblast-like,Spindle-shaped (long),Monolayer adherent growth |
| Sharing mode | Public welfare sharing |
1.Name:HEL299
2. No.:340264
3. growth properties : □ wall ;■ suspension ; □ semi-suspension and semi-wall
4. Growth conditions:
| culture medium | 90% RPMI-1640 |
| serum | 10% FBS(Diagnovum) |
| temperature | 37 ℃ |
| air condition | 5% CO2,95% AIR |
| growth algebra | P4-5 |
| frozen storage conditions | culture medium 50%, serum 40%, DMSO 10% |
5. composition:
| composition | specifications |
| a bottle of cells | T25 |
| cell culture and operating instructions | 1 copy |
Receiving notice:
1 Upon arrival, it is suggested to sit the cells in a incubator for about 4 hours, and then renew the media for recovery or subculture according to the cell density.
2 Collect the cells by centrifuge, re-suspend the collected cells with 10ml of complete media, transfer to fresh culture flask/vessel and cultivate overnight, and dispense into separate vials for subculture according to the cell density.
3 Clustered growth: agitate the culture flask to disperse the clustered cells and continue to recover or subculture.
Recovery and subculture procedure of cells ( under strict aseptic conditions )
1. The suspension cells is normally handled by changing half of the solution and sub-cultured in separate flasks, that is, transfer half of the suspension liquid to a fresh flask/vessel, add appropriate complete medium and culture it in the incubator; It can also be sub-cultured in separate flasks according to cell density.
2 Pay attention to the change of pH value of medium and cell density, renew the medium regularly, and repeat the subculture or cryopreservation when the cell density reaches 70%-80%.
Special attention: (if handle in public laboratory or first time to culture the cells, it is recommended to add penicillin streptomycin into the media)
1 Upon the receipt of the cells, renew the solution with fresh 10% FBS medium as soon as possible. It is not recommended to use the medium used for transportation in the original flask.
2 Please take photos and contact the technicians in time if the culture flask leaks upon the receipt of cells.
3 Any compliant on the cells, please take photos and contctat our technicians.
 Super Tube(Super Tube)
100297
Super Tube(Super Tube)
100297
 Y79(Y79)
100707
Y79(Y79)
100707
![bEnd.3[BEND3]](https://beinabncc.oss-cn-beijing.aliyuncs.com/News/d3e12572b0921eb522c87709e0880df6.jpg) bEnd.3[BEND3](bEnd.3[BEND3])
337672
bEnd.3[BEND3](bEnd.3[BEND3])
337672
 MC38(MC38)
337716
MC38(MC38)
337716
 MH7A(MH7A)
337864
MH7A(MH7A)
337864
 MADB106(MADB106)
339574
MADB106(MADB106)
339574